 
It is fast enough that the uncertainty principle $\Delta E \Delta t \leq \hbar/2$ allows energy conservation to be temporarily violated. The absorption and re-emission I describe is a very fast process called "virtual transition". The energy is added to a field propagating in the original direction, slightly delayed. But the business about phase fronts and directions still holds. The molecule lives in this state for a short period of time, and then the energy is returned to the field ("photon emission"). 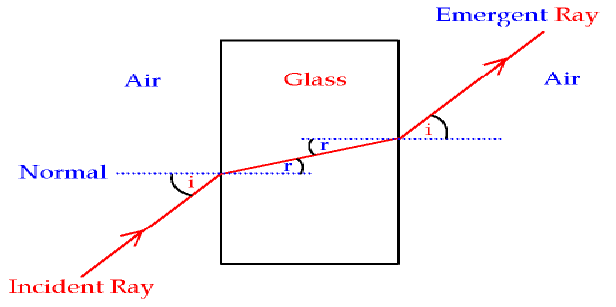
Instead of exciting the molecules into oscillation, the molecule temporarily absorbs a quantum of energy from the field (the "photon"), and the molecule is raised to an excited state. Recall that a photon is an excitation of the light field. 
But the phase fronts, and hence the direction of travel, is parallel to that of the incident wave. The re-emitted light adds to the portion of the incident light that passes unaffected. The light re-emitted from the molecules will also share that same pattern, although it will be delayed in time relative to the incident light. The incident light was traveling in a particular direction with a well-defined phase front, and so too does the pattern of oscillation in the liquid. The incident field has a particular spatial pattern, say a sine pattern, and it excites in the molecules an oscillation pattern that exactly matches. How does it know to go straight? Here's where we need all the other molecules in the liquid. After this re-emission, the light will travel at $c$. That's the classical picture of the delay experienced by light during an interaction. The molecule, then, is a radiator and can generate its own light. The energy that the field gave to the molecule will stay with the molecule for a while while this oscillation occurs. Classically, when light interacts with a molecule, the electron is set into vibrational motion. It's best to start thinking about your question in the realm of classical physics, and then modify it later to include quantum mechanics. Individual interactions, on the other hand, can occur with a particular molecule at a particular location. Like all particles in quantum mechanics, there's a chance that it could exist anywhere. It is, unfortunately, often not helpful to think of a photon as a particle that exists at a particular place in space. Next, remember that a photon is an excitation of a complete electromagnetic field. Light absorbed and re-emitted by a single molecule can go off in (almost) any direction. Light is interacting with all of the molecules. Here's a little more to help prop up the explanation.įirst, it's important to realize that in a condensed phase like a solid or liquid the light is not interacting with molecules in isolation. Yes, it's ok, but it's an explanation that has been stripped down to bare bones, and leaves out quite a bit. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
